DISRUPT PAD III Observational Study
Count on it: The DISRUPT PAD III Observational Study (OS) is the largest prospective real-world evidence for the treatment of complex, heavily calcified peripheral artery disease (PAD).


Key Findings
-
Predictable Outcomes in Challenging Situations
DISRUPT PAD III OS saw predictably consistent results across vessel beds, challenging lesions and complex patients. -
Real-world Outcomes Consistent with Randomized Trial
Shockwave IVL safely and effectively modifies calcium across multiple vessel beds. -
Optimized Procedural Insights
The use of embolic protection was less when IVL was used as the only calcium modification tool; IVL saw better results with appropriate IVL sizing (oversizing 10%).
Study Design & Characteristics
Objective:
Assess real-world peri-procedural outcomes of Shockwave Intravascular Lithotripsy (IVL) for the treatment of calcified, stenotic peripheral arteries
Design:
- Prospective
- Multicenter
- Single-blind
- Observational study
Key Inclusion Criteria:
- Rutherford classification 2-6
- Moderate-severe calcification*
- Ilio-femoral, femoral, popliteal and infrapopliteal arteries
Statistics Callout
This section presents key statistical information with numbers and descriptions.
-
1,373Complex real-world patients
-
36%Chronic limb-threatening ischemia (CLTI) patients
-
56%Diabetes mellitus
-
27%Renal deficiency
-
1,531Challenging lesions
-
90%Moderate-severe calcium**
-
115mmAverage calcified length
-
31%Chronic total occlusions (CTO)
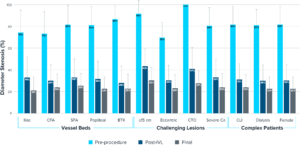
Predictable Outcomes in Challenging Situations
DISRUPT PAD III OS saw predictably consistent results across vessel beds, challenging lesions and complex patients.
Majority of Stenosis Reduction Seen from IVL Treatment
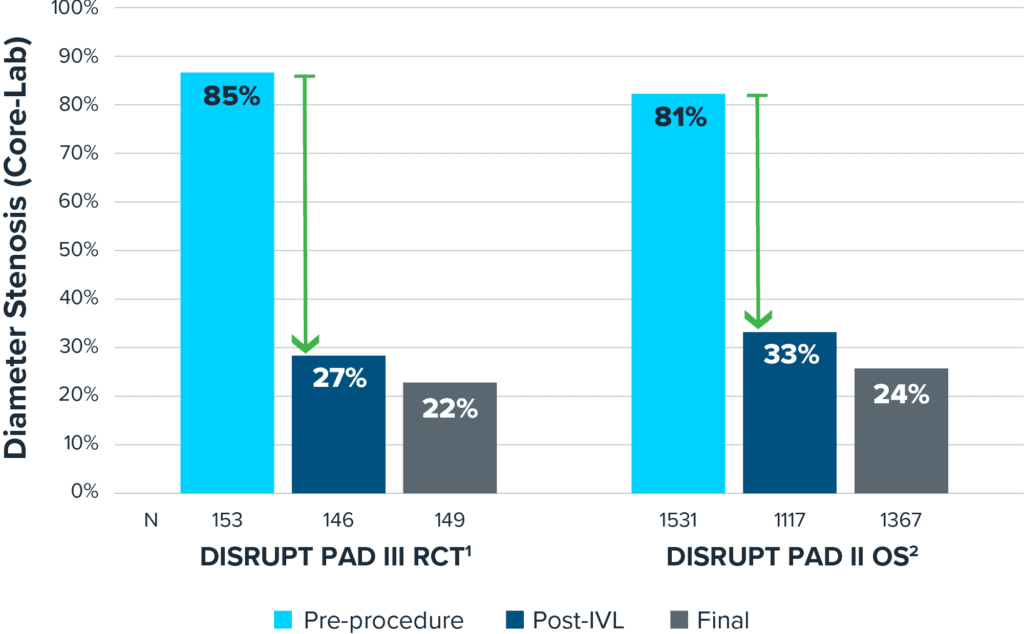
Real-world Outcomes Consistent with Randomized Trial
Shockwave IVL safely and effectively modifies calcium across multiple vessel beds.
Strong Safety Profile
| DISRUPT PAD III Randomized Clinical Trial (RCT)1 | DISRUPT PAD III OS2 | |
| N | 153 | 1,367 |
| Vessels | Superficial femoral artery (SFA)/popliteal | Iliac, common femoral artery (CFA), SFA/popliteal, infrapopliteal |
| Dissection (Type D-F) | 0% | 0.7% |
| Perforation | 0% | 0.1% |
| Embolization | 0% | 0% |
| Slow flow/no reflow | 0% | 0% |
| Abrupt closure | 0% | 0% |
| Thrombus | 0% | 0% |
Final angiographic complications (Core-Lab)
Proven Effective Calcium Modification
Shockwave IVL Procedural Insights
- Use of adjunctive technology was at the operator’s discretion
- Use of embolic protection was less when Shockwave IVL was used as the only calcium modification tool
- Shockwave IVL saw better results with appropriate Shockwave IVL sizing
When Shockwave IVL was used as the only calcium modification therapy†, there was less use of embolic protection.

Per a multivariable analysis, proper Shockwave IVL balloon sizing (≥ 1:1)‡ is an independent predictor of improved stenosis reduction but not a predictor of complications.

Conclusion
-
The DISRUPT PAD III OS 1,373 represents the largest prospective ‘real-world’ evidence for the treatment of heavily calcified peripheral artery disease.
-
The data reinforces the predictability of Shockwave IVL and its ability to consistently modify calcium across vessel beds, challenging lesions, and complex patients (CLI, dialysis & female patients).
-
Use of adjunctive technology was at the operator’s discretion – use of embolic protection was less when Shockwave IVL was used as the only the only calcium modification tool; IVL saw better results with appropriate IVL sizing.
-
Real-world outcomes of DISRUPT PAD III OS are consistent with the previously reported PAD III RCT showing that Shockwave IVL safely and effectively modifies calcium across multiple vessel beds.
Study Leadership
This section contains attributions including profile pictures, titles, descriptions, and Twitter handles.
-
Ehrin J. Armstrong, MDInterventional Cardiologist, Advanced Heart & Vein Center, Thornton, CO
-
George Adams, MDDirector of Clinical Cardioascular & Peripheral Vascular Research, UNC REX, Garner, NC
-
Sarang S. Mangalmurti, MDInterventional Cardiologist, Main Line Health, Bryn Mawr, PA
-
Nicolas W. Shammas, MDMedical Director of Cardiology Services, Trinity Bettendorf Hospital, Bettendorf, IA
-
Barry Bertolet, MDDirector of Cardiac Catheterization Laboratory NMMC, Cardiology Associates of North Mississippi, Tupelo, MS
-
William A. Gray, MDProfessor of Medicine, Main Line Health, Wynnewood, PA
-
Gunnar Tepe, MDProfessor of Radiology, Head of Diagnostic & Interventional Radiology, RoMed Clinic, Rosenheim, Germany
-
Edward Y. Woo, MDDirector of Vascular Program, Chief of Vascular Surgery, MedStar Regional, Washington D.C.
-
James F. McKinsey, MDProfessor of Vascular Surgery & Interventional Radiology, Mount Sinai Health System, New York, NY
-
Andrew Holden, MDProfessor, Director of Interventional Radiology, Auckland Hospital, Grafton, Auckland, New Zealand
-
Sahil A. Parikh, MDAssociate Professor of Medicine, Columbia University, New York, NY
-
William Bachinsky, MDCardiologist, Penn State Health Holy Spirit Medical Center, Mechanicsburg, PA
More Clinical Evidence
-
Global prospective, multi-center, single-arm study assessing the safety and effectiveness of Shockwave peripheral IVL in treating long, calcified BTK lesions.Peripheral IVL
-
The largest-ever randomized clinical study of Shockwave peripheral IVL treatment in severely calcified peripheral lesions, out to 24 months.Peripheral IVL
-
Core lab adjudicated, long-term, multi-center study exclusively enrolling heavily calcified lesions building upon DISRUPT PAD I.Peripheral IVL
*Presence of fluoroscopic evidence of calcification by PARC definition: 1) on parallel sides of the vessel and 2) extending > 50% the length of the lesion if lesion is ≥50 mm in length; or extending for minimum of 20 mm if lesion is <50 mm in length.
**PARC definition.
†Ca modifying therapy: atherectomy and/or scoring/cutting balloon.
‡PAD OS data analysis is consistent with prior PAD II data analysis and continues to support 1.1:1 sizing in the product instructions for use (IFU).
1: Tepe et al, J Am Coll Cardiol Intv 2021.
2: Armstrong EJ, Adams G, Soukas PA, Mangalmurti SS, Shammas NW, Mehrle A, Bertolet B, Gray WA, Tepe G, Woo EY, McKinsey JF, Holden A, Parikh SA. Intravascular Lithotripsy for Peripheral Artery Calcification: 30-Day Outcomes From the Disrupt PAD III Observational Study. J Endovasc Ther. 2024 Oct 18:15266028241283716. doi: 10.1177/15266028241283716. Epub ahead of print. PMID: 39422234.