COSIRA II Clinical Study Patient Information
Learn more about the COSIRA II clinical study, including the purpose of the study, who can participate, and participating site locations throughout the United Kingdom.


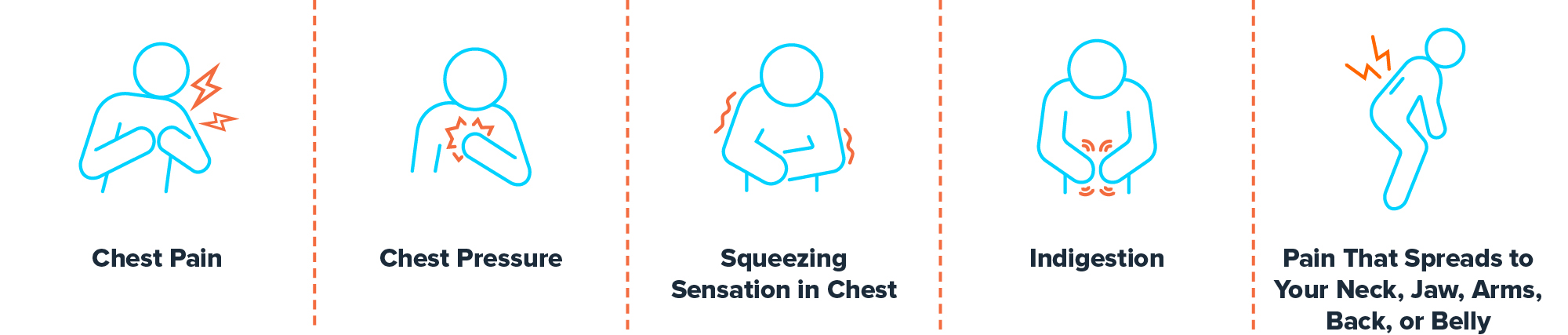
Do you suffer from chronic chest pain?
If you experience chronic chest pain despite appropriate medically-prescribed therapy and are not a candidate for another procedure, talk to your doctor about potential eligibility for participating in a study to investigate a novel treatment option.
Common symptoms of angina:
COSIRA II is a clinical study designed to evaluate the safety and effectiveness of a medical device, known as the Shockwave™ Reducer, in treating patients suffering from chronic chest pain.

The Shockwave Reducer device
The Reducer is a small, hourglass shaped tube implanted into the main vessel that collects blood once it leaves the heart muscle. The device is intended to create back pressure that increases oxygen-rich blood flow to areas of the heart not receiving normal blood flow.
Who can participate in the clinical study?
You may be able to participate in the COSIRA II study if you meet these criteria:
- You are a patient suffering from angina symptoms during ordinary physical activity such as walking at normal pace or climbing one flight of stairs despite receiving medical therapy
- You had a previous procedure (stent or surgery) that did not relieve your symptoms
- You are on several medications to control your chronic chest pain but still suffer from symptoms
What will happen if you join the COSIRA II clinical study?
If your doctor thinks you would be a good candidate for this study, you will be evaluated to see if you are eligible to participate. You may:
- Receive a study-related physical exam
- Walk on a treadmill to assess your chest pain symptoms
- Give blood for lab testing
If you participate in the study, the Reducer device may be implanted in a relatively quick procedure under local anesthesia using a catheter inserted through a small needle above your shoulder. Patients are typically discharged on the same day. You will also be required to visit the study site for follow-up visits (or will be contacted by phone) at 6 months, 12 months, 2 years, 3 years, 4 years, and 5 years to assess your symptoms, quality of life, and medications.
COSIRA II Participating Site Locations
Contact a participating site today to learn more about the COSIRA II clinical study or to be evaluated for eligibility.
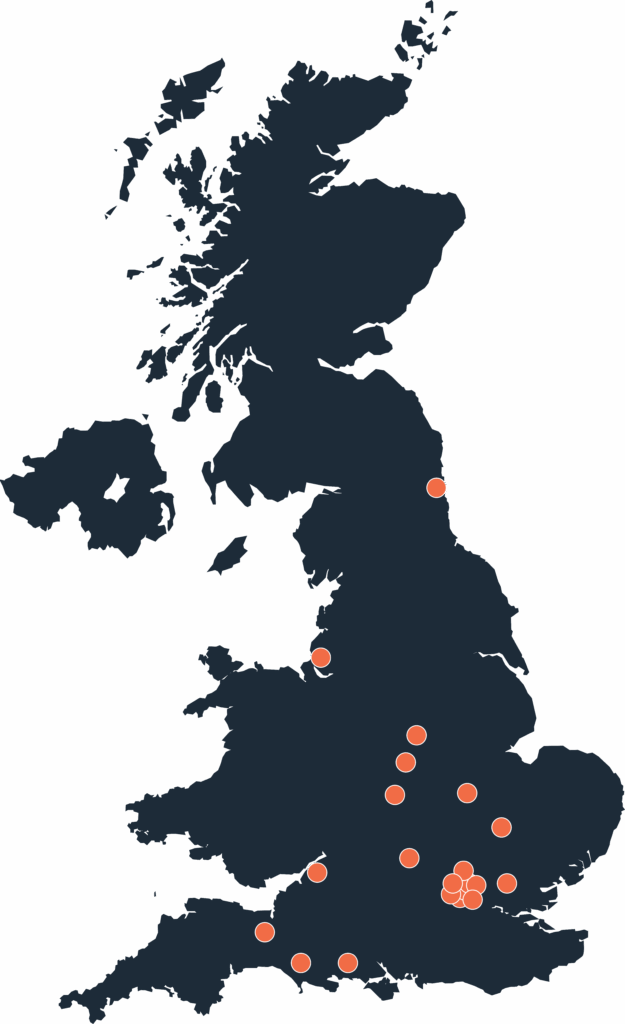
| Hospital | City | Principle Investigator |
| Imperial College | London | Dr. Rasha Al-Lamee |
| Liverpool Heart and Chest Hospital | Liverpool | Dr. Joel Giblett |
| Essex Cardiothoracic Centre | Essex | Dr. Thomas Keeble |
| Dorset County Hospital | Dorchester | Dr. Fraser Witherow |
| King’s College | London | Dr. Jonathan Byrne |
| Oxford University Hospital | Oxford | Dr. Giovanni De Maria |
| Royal Brompton Hospital | London | Dr. Ranil de Silva |
| Barts/QMUL | London | Dr. Anthony Mathur |
| Royal Free London | London | Dr. Tushar Kotecha |
| Royal Bournemouth Hospital | Bournemouth | Dr. Peter O’Kane |
| Musgrove Park Hospital | Taunton | Dr. Mohammad Sahebjalal |
| Freeman Hospital | Newcastle | Dr. Billal-Reshad Bawamia |
| Kettering General Hospital | Kettering | Dr. Prashanth Raju |
| Royal Papworth Hospital | Cambridge | Dr. Stephen Hoole |
| Bristol Royal Infirmary | Bristol | Dr. Ioannis Felekos |
| St. Thomas Hospital | London | Dr. Tiffany Patterson |
| St. George’s Hospital | London | Dr. James Spratt |
| Nottingham University Hospital | Nottingham | Dr. Andrew Vanezis |
| Queen Elizabeth Hospital | Birmingham | Dr. Alex Zaphiriou |
| Glenfield Hospital | Leicester | Dr. Bhavik Modi |