Shockwave L6 IVL in the Iliacs Discussion & Case Review
Dr. Shivani Kumar | Tufts Medical Center | Boston, MA
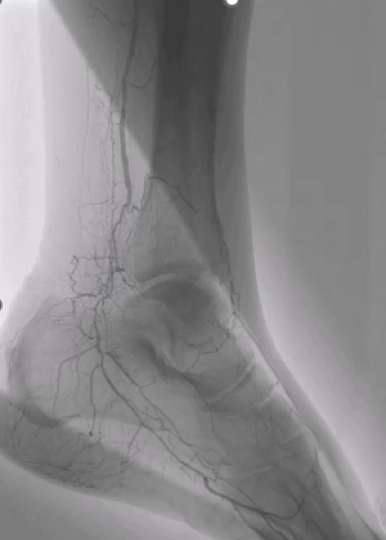
Dr. Shivani Kumar, vascular surgeon, discusses her experience treating calcified iliacs and how Shockwave IVL is uniquely positioned to treat this vessel bed.
Dr. Kumar then reviews two cases where she used Shockwave L6 to treat the left common iliac artery for femoral-femoral bypass inflow and bilateral Shockwave L6 with kissing iliac stents.
Dr. Kumar is a paid consultant for Shockwave Medical.
Peripheral IVL
Shockwave M5+, Shockwave M5, Shockwave S4, Shockwave L6 and Shockwave E8 Safety Information
In the United States: Rx only.
Indications for Use—The Shockwave Medical Intravascular Lithotripsy (IVL) System is intended for lithotripsy-enhanced balloon dilatation of lesions, including calcified lesions, in the peripheral vasculature, including the iliac, femoral, ilio-femoral, popliteal, infra-popliteal, and renal arteries. Not for use in the coronary, carotid or cerebral vasculature.
Contraindications—Do not use if unable to pass 0.014″ (M5, M5+, S4, E8) or 0.018″ (L6) guidewire across the lesion-Not intended for treatment of in-stent restenosis or in coronary, carotid, or cerebrovascular arteries.
Warnings—Only to be used by physicians who are familiar with interventional vascular procedures—Physicians must be trained prior to use of the device—Use the generator in accordance with recommended settings as stated in the Operator’s Manual.
Precautions—use only the recommended balloon inflation medium—Appropriate anticoagulant therapy should be administered by the physician—Decision regarding use of distal protection should be made based on physician assessment of treatment lesion morphology.
Adverse effects–Possible adverse effects consistent with standard angioplasty include–Access site complications–Allergy to contrast or blood thinner–Arterial bypass surgery—Bleeding complications—Death—Fracture of guidewire or device—Hypertension/Hypotension—Infection/sepsis—Placement of a stent—renal failure—Shock/pulmonary edema—target vessel stenosis or occlusion—Vascular complications. Risks unique to the device and its use—Allergy to catheter material(s)— Device malfunction or failure—Excess heat at target site.
Prior to use, please reference the Instructions for Use for more information on indications, contraindications, warnings, precautions and adverse events. www.shockwavemedical.com/ifu