Shockwave E8
Above and beyond for above and below: This Shockwave peripheral Intravascular Lithotripsy (IVL) workhorse is purpose-built to extend your capabilities when treating long calcified lesions above and below the knee.



Purpose-Built to Treat Challenging Calcium Above and Below the Knee
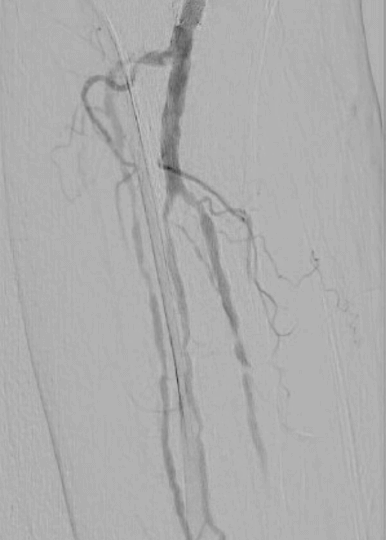
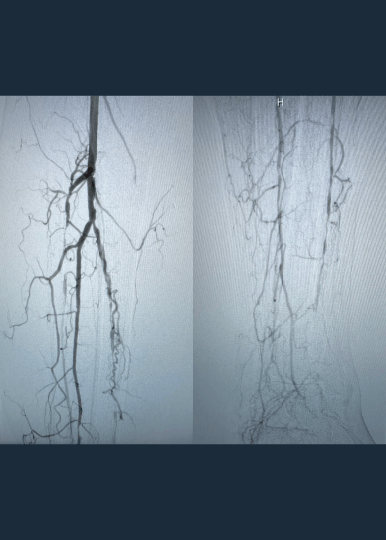
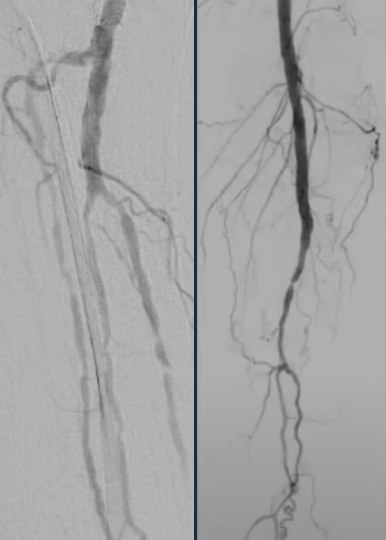
Calcified peripheral arterial disease has met its match in the new Shockwave E8 IVL catheter – our longest catheter yet. Designed for versatility, Shockwave E8’s sonic pressure waves provide a low-risk option that effectively cracks calcium where it counts, simplifying complex procedures and helping restore peripheral blood flow.
- Treats long lesions efficiently: 400 pulses at 2 Hz pulsing speed allow for efficient treatment of over 30 cm lesions
- Provides extended reach below the knee: 150 cm working length allows for the treatment of difficult-to-reach lesions
- Enables access options with sheath compatibility: the 5 Fr and 6 Fr compatibility enable the use of access sites such as pedal and radial
Shockwave E8 Specifications
| Catalog Number | Balloon Diameter (mm) | Balloon Length (mm) | Sheath Compatibility (Fr) | Working Length (cm) | Pulses/Cycles | Cycles | Pulses (max) | Balloon Crossing Profile |
| E8IVL025080 | 2.5 | 80 | 5 | 150 | 40 | 10 | 400 | .050 |
| E8IVL030080 | 3.0 | 80 | 5 | 150 | 40 | 10 | 400 | .051 |
| E8IVL035080 | 3.5 | 80 | 5 | 150 | 40 | 10 | 400 | .053 |
| E8IVL040080 | 4.0 | 80 | 5 | 150 | 40 | 10 | 400 | .055 |
| E8IVL050080 | 5.0 | 80 | 6 | 150 | 40 | 10 | 400 | .061 |
| E8IVL060080 | 6.0 | 80 | 6 | 150 | 40 | 10 | 400 | .066 |
Dr. Foteh is a paid consultant of Shockwave Medical
Disrupt PAD III Observational Study
The largest prospective real-world evidence for the treatment of complex, heavily calcified PAD
Statistics Callout
This section presents key statistical information with numbers and descriptions.
-
1,373Patients
-
1,531Lesions
-
30Sites
-
3Countries