DISRUPT CAD Pooled
Patient-pooled analysis of the global DISRUPT CAD I-IV clinical program demonstrates consistently high rates of procedural success with low rates of post-IVL procedural angiographic complications of severely calcified coronary lesions treated with Shockwave IVL.


Key Findings
-
Shockwave IVL facilitates stent delivery with low residual stenosis
High acute gain, procedural success & successful stent delivery with low residual stenosis demonstrate effective calcium modification with Shockwave IVL -
Low rates of post-IVL and post-stent procedural complications
The only Wavemaker with clinical evidence demonstrating low rates of procedural angiographic complications immediately following Shockwave IVL -
Consistent global outcomes
No statistical difference in procedural success (p=0.84) or 30-day MACE (p=0.56) across geographies studied within the DISRUPT CAD I-IV clinical program
Study Design & Characteristics
Objective:
To assess the cumulative safety and effectiveness of the Shockwave Medical Coronary IVL System for the treatment of severely calcified lesions in coronary arteries
Key inclusion criteria:
- Presence of fluoroscopic calcium visible on both sides of the arterial wall without cardiac motion
- Total calcified segment ≥ 15 mm that extends into the target lesion
- Intravascular imaging showing a calcium arc ≥ 270° in at least one cross-section
Primary safety endpoint:
Freedom from major adverse cardiovascular events (MACE) at 30 days
Primary effectiveness endpoint:
Procedural success, defined as stent delivery with residual in-stent stenosis ≤30% as assessed by the angiographic core laboratory and without in-hospital MACE
-
 Australia
Australia
-
 Belgium
Belgium
-
 Denmark
Denmark
-
 France
France
-
 Germany
Germany
-
 Italy
Italy
-
 Japan
Japan
-
 Netherlands
Netherlands
-
 Spain
Spain
-
 Sweden
Sweden
-
 United Kingdom
United Kingdom
-
 United States
United States
Statistics Callout
This section presents key statistical information with numbers and descriptions.
-
628Patients
-
72Sites
-
12Countries
Statistics Callout
This section presents key statistical information with numbers and descriptions.
-
97%Severe calcium
-
42mmCalcium length
-
99%Shockwave IVL crossing & therapy delivery
-
12%Residual stenosis*
-
92.4%Procedural success
-
99%Successful Shockwave IVL delivery
-
0.3%Serious angiographic complications*
-
1.7mmAcute luminal gain*
Low Rates of Post-IVL & Post-Stent Procedural Complications
Shockwave Medical is the only Wavemaker with published post-IVL procedural outcomes demonstrating the first-in-class safety of Shockwave IVL.
Event Rate Across the DISRUPT CAD Studies
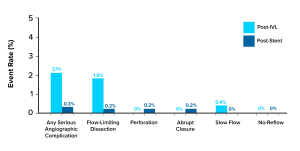
Immediately following IVL treatment (n = 561) and post-stent (n = 628) demonstrated a low rate of flow-limiting dissections (grade D or greater) with no perforation, abrupt closure, or no-reflow events following IVL treatment. Core laboratory assessed.
Shockwave IVL Facilitates Stent Delivery with Low Residual Stenosis Within Severe Calcium
Shockwave IVL increased minimal luminal diameter (MLD) and improved diameter stenosis immediately following therapy.
Procedural Angiographic Outcomes Following IVL Treatment
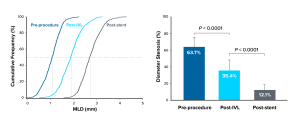
Core laboratory-assessed minimum luminal diameter cumulative frequency curves demonstrate an increase in minimal lumen diameter (MLD) immediately following Intravascular Lithotripsy (IVL) treatment with further increase post-stent. Core laboratory-assessed diameter stenosis demonstrates a significant decrease in diameter stenosis immediately following IVL treatment (p<0.0001) and post-stent (p<0.0001). For MLD and diameter stenosis measurements, pre-procedure n=625, post-IVL n=555, and post-stent n=625. Post-stent assessments of MLD and diameter stenosis include post-dilation in 94.1% of patients.
Consistently High Procedural Success & Low 30-day MACE Globally
The DISRUPT CAD Pooled analysis saw no statistical difference in procedural success and 30-day MACE across geographies. 30-day MACE was driven by non-Q-wave myocardial infarction (MI).
Safety & Effectiveness Across the DISRUPT CAD Studies
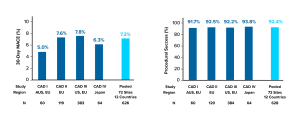
DISRUPT CAD major adverse cardiovascular event (MACE) rates at 30 days and procedural success, defined as successful stent delivery with in-stent residual stenosis ≤30% (core laboratory assessed) without in-hospital MACE, demonstrated consistent outcomes among the individual DISRUPT CAD studies. Heterogeneity among studies was evaluated using logistic regression with study as a fixed effect. All p values were not statistically significant, indicating consistency across the four studies for 30-day MACE (p = 0.56) and procedural success (p = 0.84). AUS = Australia; EU = Europe; US = United States.
More from the DISRUPT CAD Studies
-
Shockwave coronary study demonstrating the safety and effectiveness of Shockwave IVL within a Japanese patient population.Coronary IVL
-
Largest and most rigorous Shockwave study showcasing the safety, effectiveness and ease of use of Shockwave coronary IVL .Coronary IVL
-
European post-market study confirming DISRUPT CAD I results and showing strong safety and procedural success with Shockwave coronary IVL.Coronary IVL
*Procedural angiographic outcomes post stent. Independent angiographic core lab assessment.
Kereiakes et al. Intravascular Lithotripsy for Treatment of Calcified Coronary Lesions: Patient-Level Pooled Analysis of the Disrupt CAD Studies. JACC Cardiovasc Interv. 2021 Jun 28;14(12):1337-1348.